Revised: 25 March 2013
Medicines
Over-the-counter (OTC) medicines business reforms: Staged implementation plan
Introduction and overview
The Over-the-counter (OTC) Medicines Business Process Reform consultation paper developed to support the introduction of a new OTC medicine business process noted that some aspects could be initiated immediately, whereas other aspects would be introduced over time.
In response many submitters requested more information about the staged implementation and specifically requested dates for planning purposes. Submitters also called for longer implementation in either Australia or New Zealand depending on the degree of change to current regulatory practices.
The accompanying timeline has been developed to clearly communicate the staged implementation as it applies in Australia and New Zealand. The timeline demonstrates that each regulator is seeking to harmonise the business processes for receiving and assessing new OTC medicine applications and that further harmonisation will be undertaken progressively during the establishment of the Australian and New Zealand Therapeutic Products Agency (ANZTPA).
Staged Implementation Plan
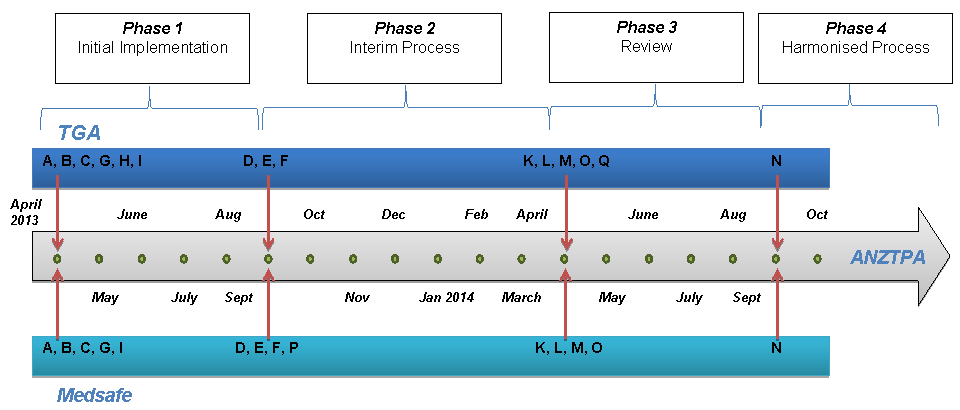
| Milestones | |
|---|---|
| A | Applications expected in Common Technical Dossier (CTD) format as described by the International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH)1 |
| B | Non-CTD dossiers must be cross-referenced to CTD format for ease of evaluation. Non-compliant dossiers will be returned and require re-submission. |
| C | Application category must be identified. If incorrect category is selected, applicant is given an opportunity to correct. |
| D | All applications in CTD format. Incorrectly constructed applications will be returned and will require re-submission (no forfeit of fee). Screening will re-start upon re-submission. |
| E | Applications submitted incorrectly into a lower category will be returned (no forfeit of fee). Screening will start upon re-submission. |
| F | Medsafe and TGA to review:
|
| G | All applications screened for completeness.2 Applications are not accepted unless they are complete and adhere to the relevant guidelines (ARGOM, NZRGM/GRTPNZ). Incomplete applications will be returned and will require re-submission. |
| H | Monograph trial commences with progressive release of draft monographs for consultation. As each monograph is finalised applications may be submitted through the N2 route (Australia only). |
| I | Medsafe aim to achieve ANZTPA aspirational timelines. TGA will
aim to achieve timelines as specified in the consultation document. A maximum of two rounds of requests for information (RFI) and no new data unless in response to a specific request from the regulator. |
| K | Performance targets reviewed. TGA introduces a target timeline
for the initial evaluation. Interim review of monograph route. |
| L | Medsafe and TGA commence review and harmonisation of changes to medicines and categorise changes according to the inherent risk. |
| M | CTD format and correct category of applications mandatory. Incorrect and incomplete applications will be returned and any application fee forfeited. |
| N | Review of OTC processes, timelines and key performance indicators (KPIs). |
| O | Medsafe and TGA commence review and harmonisation of data requirements for N4 category applications. |
| P | Medsafe undertakes a review of monograph trial with the view to introducing monographs in NZ if feasible. |
| Q | TGA completes transition to paperless applications. |
- The TGA require a paper copy and electronic copy (CD, DVD or data stick) must be provided. Medsafe require a paper copy and two electronic copies (CD, DVD or data stick must be provided.
- The TGA will collect application fees prior to screening and issue effective letters if application is complete. Medsafe will screen upon lodgement and if complete an invoice will be issued.
Key points in implementation timeline:
Application formats and administrative processes are common in both jurisdictions from 15 April 2013. There will be a transitional period of 12 months to allow applicants to become familiar with expected formats.
Guidance to assist applicants is available. Sponsors are encouraged to provide feedback and the guidance will be progressively updated during the transitional period to improve clarity and understanding.
Both regulators will implement target timelines for the administrative steps with review and modification of the timelines to be undertaken during an 18 month period. Both regulators will monitor and report performance against target timeframes to introduce predictability and transparency for applicants.
The monograph route to approval will be trialled by the Australian Therapeutic Goods Agency (TGA) to determine the costs and benefits to the regulator and uptake by applicants. During the trial period further monographs will be developed to expand the number of eligible products.
Medsafe will monitor the trial and introduce monograph if feedback is positive.
Harmonisation of the data requirements for OTC medicine applications and categorisation of changes to existing medicines will continue during the lead up to ANZTPA to enable a fully harmonised and integrated regulatory framework to commence in July 2016.





