Published: July 1998
![]()
Publications
Top Ten Adverse Events to Sumatriptan in the IMMP
Prescriber Update 16: 37–40
July 1998
Dr David Coulter
Director IMMP
Dunedin
Adverse reactions to sumatriptan are common, but usually minor and transient, and many patients find them acceptable because of the rapid relief they get from their migraine. In order to avoid alarm it is wise to inform patients prescribed sumatriptan for the first time that unusual symptoms may occur. Practitioners should be aware that sumatriptan is contraindicated in the presence of ischaemic heart disease and Prinzmetal’s angina, and that it should not be co-prescribed with ergotamine preparations.
Sumatriptan (Imigran), a medicine used in the acute treatment of migraine and cluster headache, has been monitored in the IMMP since marketing began in April 1991. Being an expensive medicine, its growth in the market was slow until the cost of the injection was partially subsidised in September 1992. Imigran injection was fully subsidised in April 1996, but the patient must meet the full cost of the tablets.
As at 30 June 1997 there were 10608 patients in the IMMP cohort, 7966 (75%) of whom were women. The age and gender distribution is shown in the graph. The peak frequency of use for all patients is in the 40-49 year age group (33.8%), and the ratio of women to men is much greater around the years of the menopause. Over the whole period, twice as many patients have used the injections as have used the tablets.
Age groups (at first prescription) and gender of patients on sumatriptan
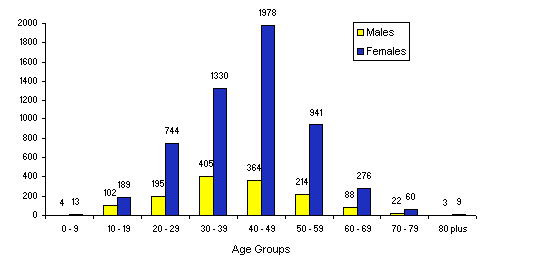
16% of sumatriptan users reported an adverse event; most minor
2828 adverse events have been reported from 1678 (15.8%) patients. The term ‘events’ is used, rather than reactions, as it is uncertain whether some common events (e.g. nausea and fatigue) are due to the disorder being treated or to the medication. The reporting rate for injections is twice that of the tablets (17% versus 8.8%) and for women it is 1.6 times greater than for men (17.5% versus 11.2%).
The 10 most frequently reported adverse events to sumatriptan
Number of patients (rate/1000 patients)
| Adverse event | Injections (n=7875) |
Tablets (n=3990) |
Rate Ratio injection/tablet |
|---|---|---|---|
| Sensory disturbance | 295 (37.5) | 48 (12.0) | 3.1 |
| Nausea/vomiting | 203 (25.8) | 96 (24.1) | 1.1 |
| Headache complications | 198 (25.1) | 42 (10.5) | 2.4 |
| Fatigue/malaise/somnolence | 185 (23.5) | 77 (19.3) | 1.2 |
| Chest pain | 178 (22.6) | 36 (9.0) | 2.5 |
| Injection site reaction | 139 (17.7) | ||
| Dizziness/syncope | 121 (15.4) | 28 (7.0) | 2.2 |
| Cardiac dysrhythmia | 88 (11.2) | 19 (4.8) | 2.3 |
| Throat tightness | 72 (9.1) | 7 (1.8) | 5.1 |
| Hot & cold feelings / shivering / sweating | 71 (9.0) | 16 (4.0) | 2.3 |
Sensory disturbances most frequently reported adverse event
The table above lists the ten most commonly reported adverse events to sumatriptan. Sensory disturbance of the skin was the most frequent. There were a variety of effects with paraesthesiae being the most common, followed by numbness, a burning feeling and hypersensitivity to heat (e.g. in the shower). Often the sensation was localised to the scalp, neck or one limb, but more often it appeared generalised. Not included in the figures were reports of similar sensations in the mouth, throat and nose.
Nausea/vomiting most likely due to migraine not sumatriptan
Nausea and/or vomiting were the second most frequently reported adverse events. Unlike most other events, the rates for injections and tablets were similar suggesting nausea or vomiting is due to the condition and not the medicine. However, there were a few reports where the patient stated that the nausea or vomiting only occurs, or is worse, with sumatriptan. There were three reports where the patient had nausea or vomiting with tablets, but not the injection, and two of the reverse situation. It is likely that most episodes of nausea or vomiting are due to the migraine. A similar situation applies to the complaints of fatigue, malaise and somnolence where the rates for injections and tablets were also similar.
Recurrence of headache common due to short half life
Some patients report worsening of the headache or an unpleasant change in its character. This usually lasts for 10 to 30 minutes, but may continue for several hours. Others said the headache recurs within a few hours of initial relief and a second dose is needed after which the migraine usually subsides. Sumatriptan is short-acting, with a plasma half life of only two hours, and recurrence of the headache is common. One specialist migraine clinic gives a frequency of recurrence with subcutaneous sumatriptan of 53%.1 In a number of reports the recurrence seems like a rebound phenomenon which develops quickly and more severely than the initial attack.
Contraindications includes IHD & Prinzmetal’s angina
Chest pain or tightness is a common complaint with sumatriptan, occurring rapidly after injection with most episodes lasting 5 - 30 minutes, but sometimes for several hours. The IMMP data show that throat tightness is often linked with chest pain or tightness.
The reason for these events is unknown, however, sumatriptan is a potent vasoconstrictor and may cause some vasospasm of the coronary arteries. It is therefore contraindicated in patients with a history of ischaemic heart disease (IHD) or Prinzmetal’s angina, and should not be used in patients with risk factors for IHD without prior evaluation for underlying cardiovascular disease. Where cardiovascular disease is absent, and a trial of sumatriptan prescribed, the first dose should be given under supervision. There are a few reports in the literature of myocardial infarction, some fatal, in patients with known or unsuspected coronary artery disease taking sumatriptan. There are no such reports in New Zealand, but there are two reports of angina.
Sumatriptan is also contraindicated for 24 hours after using ergotamine preparations, and ergotamine should not be used within six hours after using sumatriptan. The combination may be strongly vasospastic.
No serious cardiac dysrhythmias reported
The great majority of reports of cardiac dysrhythmia are described as tachycardia or palpitations. No serious problems have been reported. These may reflect an autonomic disturbance which is also likely to be responsible for the episodes of dizziness and syncope or hot and cold feelings, shivering or sweating.
Some patients have experienced strange feelings, such as depersonalisation, after sumatriptan use. Patients should be warned of this possibility.
In general, adverse reactions to the use of sumatriptan are common but minor, and most last for only a very short period of time.
Correspondence to Dr David Coulter, Director IMMP, PO Box 913, Dunedin. phone 03 479 7249, fax 03 477 0509, e-mail david.coulter@stonebow.otago.ac.nz
Reference
- Dalhof C. Headache recurrence after subcutaneous sumatriptan and early treatment. Lancet 1992;340:909.





